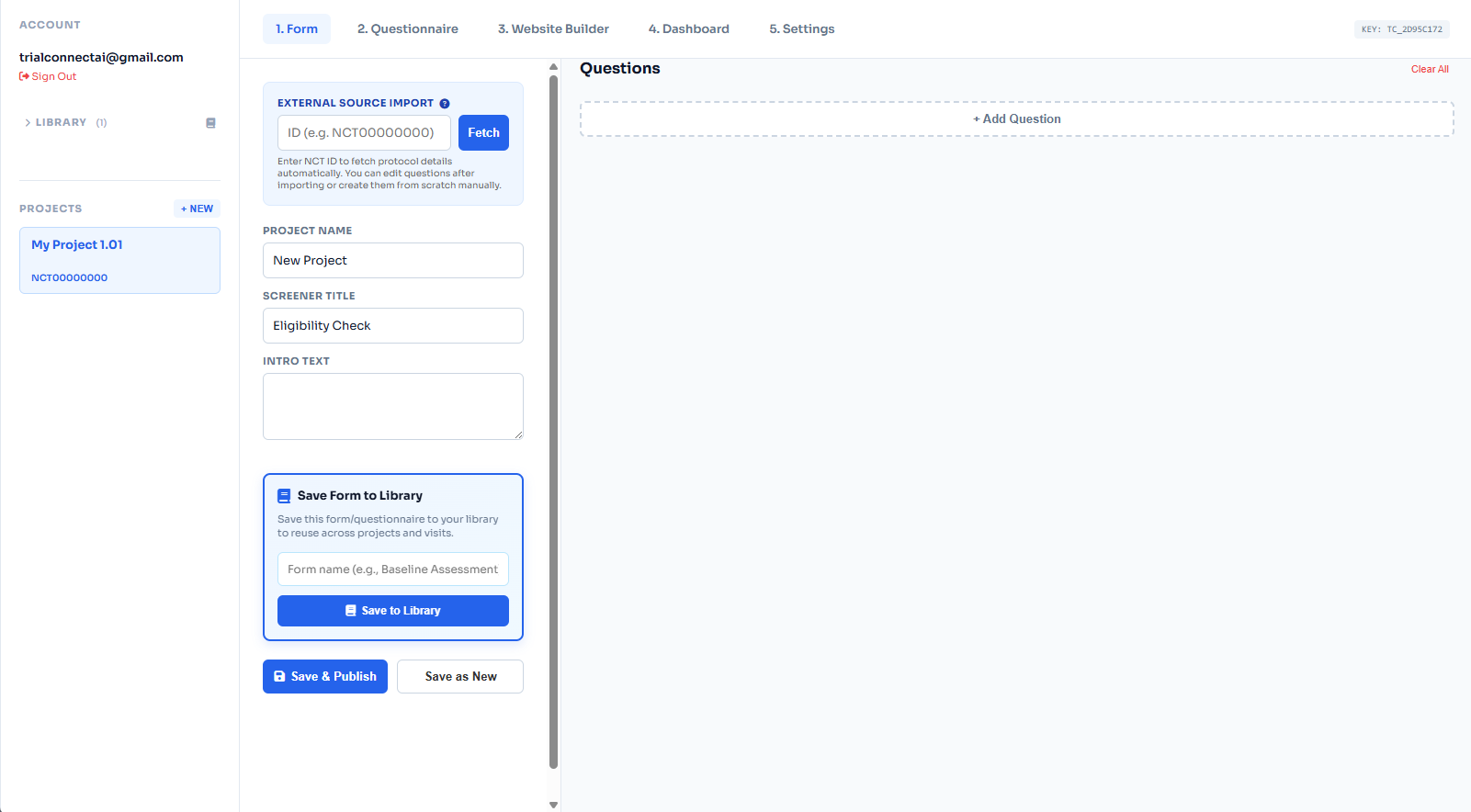
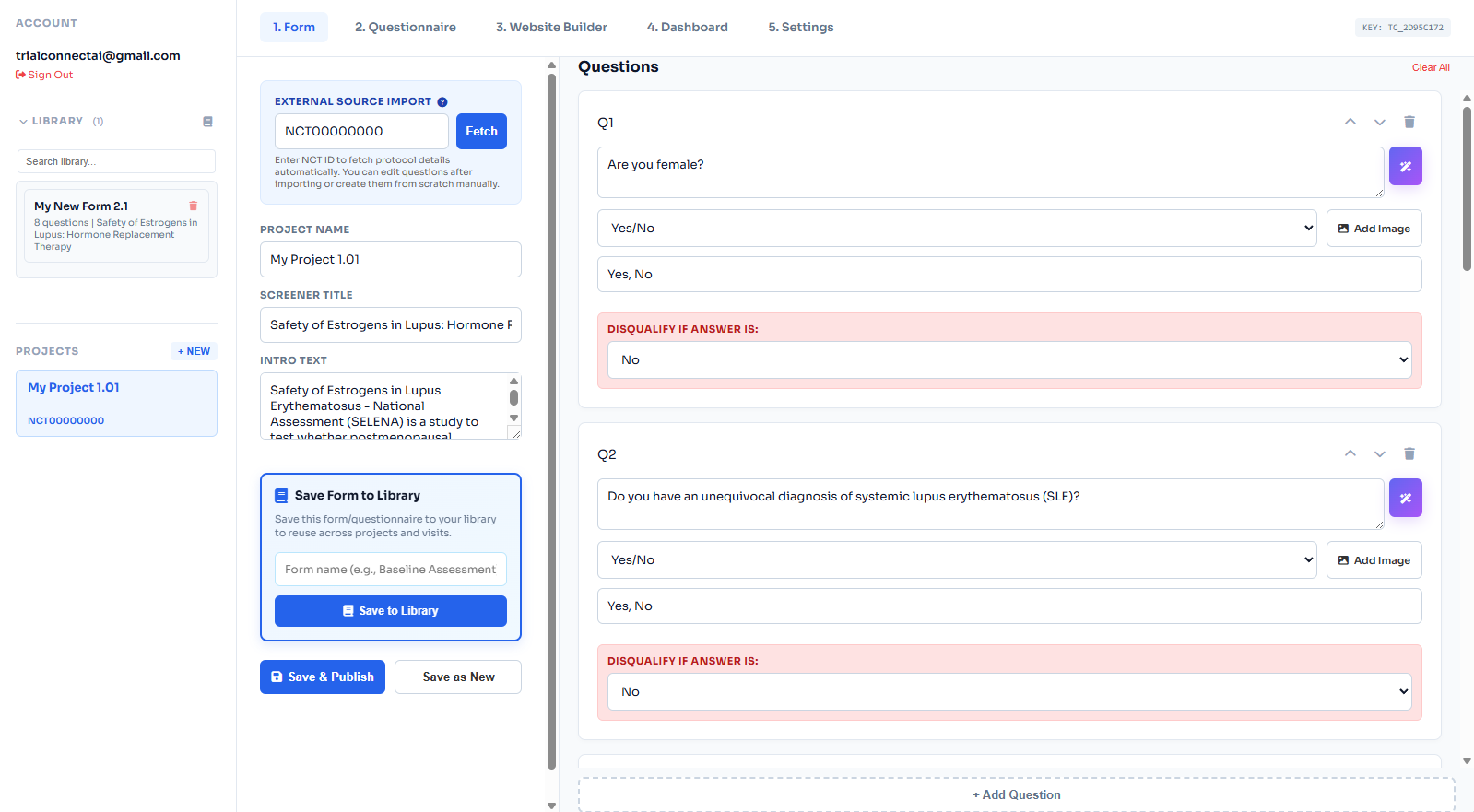
Patient Prescreener Software for Clinical Trials
Collect eligibility and intake details, then route qualified patients to your study team.
Eligibility First
Capture inclusion and exclusion criteria early to reduce coordinator workload.
Patient Intake
Collect intake details with mobile-first, patient-friendly forms.
Recruitment Ready
Launch prescreeners via embedded forms, QR codes, and study microsites.
Built for clinical regulatory compliance and security
Screener Studio is designed for clinical research teams who need reliable, secure prescreening workflows.
Consent Capture
Collect consent and contact details as part of the prescreener flow.
Audit Logging
Maintain an audit trail to support clinical operations.
Secure Storage
Protect patient data with secure workflows and access controls.
Prescreener FAQs
How long does it take to launch a prescreener?
Most teams can publish a prescreener once eligibility criteria are defined.
Can prescreeners be embedded on hospital sites?
Yes. Use embedded forms or buttons that open the prescreener directly.
How are submissions managed?
Coordinators can review submissions, track eligibility status, and manage follow-up.
Does the prescreener support study-specific branding?
Yes. Each study can use its own branding and deployment options.